How to read the Periodic Table?
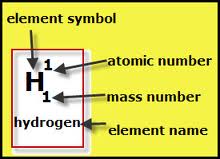
Each element is given it's own square in the Periodic Table grid.
Each element has it's own unique, personality that is made up of:
1: the element symbol
2: the element name
3: their atomic number
4: and their mass number.
The element symbol is just a shortening of the element name. In the example above, Hydrogen is shortened to 'H'.
What is the Atomic Number?
An atomic number is the number of protons in the atom's nucleus (centre). Each element has a unique number that identifies how many protons
are in a single atom. For example, all Hydrogen atoms contain one proton so the atomic number is '1'.
What is the Atomic Mass?
The Atomic Mass is a number that describes the size/weight of an atom. On this page you will learn how to interpret the personality of each element on the periodic table.
In the example above the element is Hydrogen and the diagram explains how to read the element. Just to re-cap, the atomic number is how many protons are in the atom. The mass number is how many protons and neutrons are in the atom.
The element symbol is usually just a shortening of the element's name or a code to describe the element. Generally you won't see the element name on the periodic table because it is too long to insert. If the atomic number and mass number are different it means that the element is unstable. Elements that are unstable are highly radioactive.
How do you know if an element is unstable?
Atoms that have an equal number of protons and neutrons are stable. In most atoms the protons and the neutrons found in the nucleus are held together strongly. The nucleus of these atoms are called stable. However in some atoms the neutrons and protons are not held together strongly and these are called unstable. When a nucleus is unstable neutrons are removed and this is commonly called radioactivity.
Uranium is the best known radioactive element as it is used in atomic bombs.
Reading the Periodic Table
Every element in the first column (group one) has one electron in its outer shell. Every element in the second column (group two) has two electrons in the outer shell. As you keep counting the columns, you'll know how many electrons are in the outer shell. There are some exceptions to the order when you look at the transition elements, but you get the general idea.
http://www.chem4kids.com/files/elem_transmetal.html
Each element has it's own unique, personality that is made up of:
1: the element symbol
2: the element name
3: their atomic number
4: and their mass number.
The element symbol is just a shortening of the element name. In the example above, Hydrogen is shortened to 'H'.
What is the Atomic Number?
An atomic number is the number of protons in the atom's nucleus (centre). Each element has a unique number that identifies how many protons
are in a single atom. For example, all Hydrogen atoms contain one proton so the atomic number is '1'.
What is the Atomic Mass?
The Atomic Mass is a number that describes the size/weight of an atom. On this page you will learn how to interpret the personality of each element on the periodic table.
In the example above the element is Hydrogen and the diagram explains how to read the element. Just to re-cap, the atomic number is how many protons are in the atom. The mass number is how many protons and neutrons are in the atom.
The element symbol is usually just a shortening of the element's name or a code to describe the element. Generally you won't see the element name on the periodic table because it is too long to insert. If the atomic number and mass number are different it means that the element is unstable. Elements that are unstable are highly radioactive.
How do you know if an element is unstable?
Atoms that have an equal number of protons and neutrons are stable. In most atoms the protons and the neutrons found in the nucleus are held together strongly. The nucleus of these atoms are called stable. However in some atoms the neutrons and protons are not held together strongly and these are called unstable. When a nucleus is unstable neutrons are removed and this is commonly called radioactivity.
Uranium is the best known radioactive element as it is used in atomic bombs.
Reading the Periodic Table
Every element in the first column (group one) has one electron in its outer shell. Every element in the second column (group two) has two electrons in the outer shell. As you keep counting the columns, you'll know how many electrons are in the outer shell. There are some exceptions to the order when you look at the transition elements, but you get the general idea.
http://www.chem4kids.com/files/elem_transmetal.html
Periods
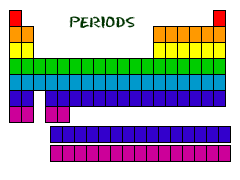
Periods are the rows in a periodic table that go left to right. Don't get confused with the groups which go vertically. Even though they may skip some squares in between. When you analyse the periodic table, each of the of rows is considered to be a different periodic (Like PERIODic table.) In the periodic table, if they are in the same row (period) it means that they have common properties. For example, every element in the top row (the first period) has one orbital for its electrons. If you want to learn about what orbitals are they are explained in the next section.
It is the same for the second row as they have two orbitals for their electrons. It goes down the periodic table like that. At this time, the maximum number of electron orbitals or shells for any element is seven. All of the elements in a period have the same number of atomic orbitals.
It is the same for the second row as they have two orbitals for their electrons. It goes down the periodic table like that. At this time, the maximum number of electron orbitals or shells for any element is seven. All of the elements in a period have the same number of atomic orbitals.
Groups
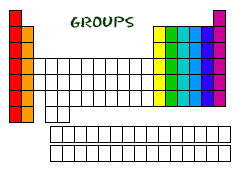
Now to Groups! Since you know about the rows the periodic table also has a common name for the columns in the chart as well. Columns in the periodic table go from top to bottom and is called a group. The elements in a group have the same number of electrons in their outer shell. The outer electrons in the atom are called valence electrons. Valence elctrons are electrons of an atom, located in the outermost shell (valence shell) of the atom, that can be transferred to or shared with another atom. They are very important because they are what make the chemical bonds between other elements.
What are Orbitals?
Orbitals are derived from the word orbit, meaning the curved path through which objects move around. As you know from the atom page, electrons are always moving. They spin around the nucleus of an atom. As the electrons fly around they can move in any direction, as long as they stay in their shell. They can go upwards, downwards, sidewards, diagonally etc. Electrons are always spinning in those atomic shells and those shells (also known as orbitals) are the distances from the nucleus. If you are an electron in the first shell, you are always closer to the nucleus than the electrons in the second shell.
The Basics of Shells:
1. The center of the atom is called the nucleus.
2. Electrons are found in areas called shells. A shell is sometimes called an energy level.
3. Shells are areas that surround the center of an atom.
If you want to learn more about orbitals click on the web address below.
http://chem4kids.com/files/atom_orbital.html
The Basics of Shells:
1. The center of the atom is called the nucleus.
2. Electrons are found in areas called shells. A shell is sometimes called an energy level.
3. Shells are areas that surround the center of an atom.
If you want to learn more about orbitals click on the web address below.
http://chem4kids.com/files/atom_orbital.html













