What are Atoms?

Atoms are the basis of chemistry. They are like the ingredients in a recipe. Without the ingredients (atoms) you can’t possibly make a delicious recipe. It’s the same with a language. You need an alphabet to construct a new language otherwise it is nothing but nonsense. Atoms are the general word for matter.
Atoms consist of three different sections: Protons, Neutrons and Electrons.
A Proton is a positively charged particle in the nucleus of an atom that balances out the charge of surrounding electrons.
Neutrons are particles inside the nucleus of an atom that are neutral - they have no charge. To help, the word neutron comes from the base word neutral meaning of no particular kind. When you combine a proton and a neutron together, they create a nucleus (the center) of an atom.
Electrons are the tiny, negatively charged particle that are also part of atoms. The electrons circle around
the nucleus to equalize the electronic charge.
Atoms consist of three different sections: Protons, Neutrons and Electrons.
A Proton is a positively charged particle in the nucleus of an atom that balances out the charge of surrounding electrons.
Neutrons are particles inside the nucleus of an atom that are neutral - they have no charge. To help, the word neutron comes from the base word neutral meaning of no particular kind. When you combine a proton and a neutron together, they create a nucleus (the center) of an atom.
Electrons are the tiny, negatively charged particle that are also part of atoms. The electrons circle around
the nucleus to equalize the electronic charge.
What is the difference between an Atomic Number and a Mass Number?
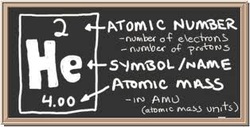
What is the Atomic Number?
An atomic number is the number of protons in the nucleus. This determines an element's atomic number. In other words, each element has a unique number that identifies how many protons are in a single atom. For example, all Helium atoms contain two protons and which is also the atomic number of 2.
The atomic number of an element will never change meaning that the number of protons in the nucleus of every atom in an element always stays the same.
What is the Atomic Mass?
The Atomic Mass is a number that describes the size of an atom. The Atomic Mass is the total mass of protons, neutrons and electrons in a single atom.
If you are interested in learning more about atomic numbers and mass numbers click the button below.
An atomic number is the number of protons in the nucleus. This determines an element's atomic number. In other words, each element has a unique number that identifies how many protons are in a single atom. For example, all Helium atoms contain two protons and which is also the atomic number of 2.
The atomic number of an element will never change meaning that the number of protons in the nucleus of every atom in an element always stays the same.
What is the Atomic Mass?
The Atomic Mass is a number that describes the size of an atom. The Atomic Mass is the total mass of protons, neutrons and electrons in a single atom.
If you are interested in learning more about atomic numbers and mass numbers click the button below.














